Diffusion PPT and PDF Report Free: Diffusion is that the net movement of molecules or atoms from a section of high concentration with a high chemical potential to a section of low concentration with low chemical potential. This is often conjointly named because of the movement of a substance downs an amount gradient. A gradient is that the amendment within the price of an amount e.g. concentration, pressure, or temperature with the modification in another variable, typically distance. An amendment in concentration over a distance is named an amount gradient, an amendment in pressure over a distance is named a pressure gradient, and an amendment in temperature over a distance could be a referred to as a gradient. The word diffusion derives from the Latin word diffundere, which suggests “spreading out”. A substance that “spreads out” is moving from a section of high concentration to a section of low concentration.
Diffusion PPT and PDF Report Free
A characteristic feature of diffusion is that is dependent on particle stochastic process, and ends up in combining or mass transport while not requiring directed bulk motion. Bulk motion, or bulk flow, is that the characteristic of temperature change. The term convection is employed to explain the mixture of each transport phenomena. Diffusion may be a physical method wherever molecules of a cloth move from a region of high concentration i.e. the region which has more molecules to a part of low concentration i.e. the region which has fewer molecules. Diffusion will solely work with liquids and gases. Here are some samples of diffusion:
- The smell of ammonia spreads from the back of the schoolroom to the front area.
- Food coloring born on the beaker spreads out.
- The smell of food spread within the total house.
Molecules tend to maneuver from regions of high concentration to places of low concentration, simply by moving haphazardly. As an example, there’s a lot of oxygen during a respiratory organ than the presence of oxygen in the blood thus oxygen molecules can tend to maneuver into the blood. Similarly, there are a lot of CO2 molecules in the blood than within the respiratory organ thus CO2 molecules can tend to maneuver into the respiratory organ. It happens in cell biology, wherever little molecules merely diffuse via the cell wall, however, larger molecules solely get through by victimization energy.
Factors which affect diffusion:
The factors which affect diffusion are explained with the help of a diagram:
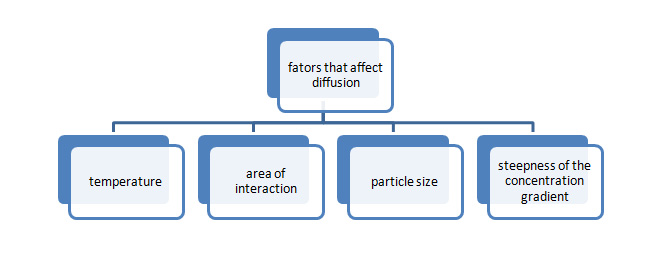
Fig1: affecting factors of diffusion
Diffusion is affected by temperature, space of interaction, steepness of the concentration gradient and particle size. Every one of those factors severally and jointly will alter the speed and extent of diffusion.
- Temperature: In any system, molecules are moving with a particular quantity of K.E. This is often typically not directed in any explicit manner and may seem random. Once these molecules impinge on each other, there’s a modification within the direction of movement also as changes to momentum and rate. As an example, if a block of solid (carbon dioxide in solid form) is placed within a box, CO2 molecules within the center of the block largely impinge on one another and obtain preserved at intervals the solid mass. However, for molecules within the edge, chop-chop moving molecules within the air conjointly influence their movement, permitting them to diffuse into the air. This creates an amount gradient, with a concentration of CO2 step by step decreasing with distance from the lump of solid.
- Area of interaction: This property is often ascertained even higher if the gas has an odor or color. As an example, once iodine is sublimated over a hot stove, purple fumes begin to see and blend with air. If sublimation is dispensed during a slender vessel, the fumes diffuse slowly out towards the mouth of the instrumentation and so quickly disappear. Whereas they’re confined to the smaller area among the melting pot, the speed of diffusion remains low.
- The steepness of concentration gradient: Since diffusion is supercharged primarily by the likelihood of molecules moving far from a section of upper saturation, it straightaway follows that once the medium (or solvent) includes a terribly low concentration of the matter, the likelihood of a molecule diffusive far from the central area is higher.
- Particle size: At any given temperature, the diffusion of a smaller particle is a lot of fast than that of a larger-sized molecule. This is often associated with each the mass of the molecule and its extent. A heavier molecule with a bigger extent can diffuse slowly, whereas smaller, lighter particles can diffuse a lot of quickly. For instance, whereas element gas can diffuse slightly a lot of quickly than CO2, each of them can move a lot of quickly than iodine gas.
Types of diffusion:
There are two types of diffusion and those are mentioned below:
- Simple diffusion
- Facilitated diffusion
Content of the Seminar and PDF Report for Diffusion
- What is Diffusion
- Diffusion Based Process
- Applications
- Advantages
- Disadvantages
- References
Here we are giving you Diffusion Seminar and PPT with PDF report. All you need to do is just click on the download link and get it.
Diffusion PPT and Seminar Free Download
Diffusion PDF Report Free Download
It was all about Diffusion Seminar and PPT with pdf report. If you liked it then please share it or if you want to ask anything then please hit comment button.