Free Download Nanoparticles Seminar PPT and PDF Report
Free Download Nanoparticles Seminar PPT and PDF Report: Nanoparticles are the easiest form of structures which have the sizes in the range of nanometer. A nanoparticle is considered to a group of atoms which are bonded together and possess a structural radius of less than 100 nm. The nanoparticles include the following:
- Large molecules like hydrogen-bonded assemblies of water molecules and proteins
- Fullerenes
- Metal clusters
Nanoparticles are very common in nature and for example, the proteins are present in all the biological systems.
Nanoparticles Seminar PPT and PDF Report
Physically Manufactures Nanoparticles:
The difference in the nanoparticles can be made between the hard matter nanoparticles which are called as nanoclusters and the soft nanoparticles which are Bioorganic and large molecules. Now, we are going to learn about the nanoclusters:
- Nanoclusters: A nanocluster is a particle which has a size in the range of nanometer and has equal subunits. The subunits can be atoms of a single element or many molecules or even consist of atoms of many elements in subunits with equal alloys. The nanocluster has the properties which are guided by the number of subunits they have. Due to this reason, they are seen as the intermediate stage between bulk material and single atom or molecule. If once the particles are made to the size of a nanocluster then the properties may vary with every addition of atom and with every subtraction of an atom.
- Size does matter: In this, we are going to know about the different parameters which are related to the size and those are as below:
- Geometric structure
- Electronic structure
- Jellium
- Optical properties
- Reactivity
- Melting temperatures
- Magnetic properties
- Geometric structure: The crystal structure of a big nanocluster is similar to the bulk structure of a material with slightly different lattice parameters. For example, the clusters of copper tend to have a structure of FCC.
- Electronic structure: The formation and abundances of big clusters are affected by the structural magic numbers. In the case of tiny clusters, the electronic structure of complete atoms is of much importance. In the clusters, the electronic magic numbers are observed.
- Jellium: In the theoretical modeling of nanoclusters, the jellium model has been utilized. The jellium model of clusters is explained in the below diagram:
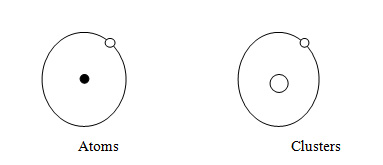
Fig1: Jellium model of Clusters
If we observe from the jellium model’s point of view, the cluster’s electronic structure changes from that of a single atom and varies greatly when compared with the bulk material. When a bulk lattice is made or formed then the energy levels of the atom which are discrete are smudged out into the energy bands. For example, the conductions bands and valence bands are present in the metals and semiconductors. As the particles reduce in the size then there will be a great change in these bands as the discrete energy levels take the place of a continuous density of states in bulk.
- Optical properties: In the semiconductors, the band gap enhances as the size of the cluster reduces. The energy level separations and the individual energy levels are also dependent on the cluster’s size. These affect the energies required for the transitions of electrons to excited states. The clusters of distinct sizes have distinct spectra of absorption and distinct colors.
- Reactivity: Reactivity is dependent on the electronic structure. The reactivity of a nanocluster is high when compared with the bulk and this due to the great surface to volume ratio. For example, the nanoclusters of gold are highly reactive.
- Melting temperatures: The clusters have low melting temperatures when compared with the bulk materials and this is because of the cluster’s high surface to volume ratio.
- Magnetic properties: The tiny clusters have interesting magnetic properties and as the size of the cluster reduces then it will be easier for them to exhibit the behavior of ferromagnetic.
- Production of clusters: There are three distinct physical methods for producing clusters and those are explained below:
- Energetic vaporization
- Seeded supersonic nozzle sources
- Gas aggregation cluster sources
- Applications of clusters: The applications of nanoclusters are explained below:
- High energy deposition
- Low energy deposition
- Surface smoothing
What are the benefits of nanoparticles?
Have a look at the advantages of nanoparticles with proper explanation below:
- Nanoparticles helpful in revolutionizing an variety of things in the manufacturing areas.
- It possess with an solid set of insulating properties for paving the way for new techniques.
Content of the Seminar and PDF Report for Nanoparticles
- Introduction
- Definition
- Classificaton Of Nanoparticles
- Advantages Of Nanoparticles
- Disadvantages Of Nanoparticles
- Preparation of polymeric Nanoparticles
- Targeted Nanoparticles
- Equipments for Nanoparticles
- Evaluation of Nanoparticles
- Conclusion
- References
Here we are giving you Nanoparticles Seminar and PPT with PDF Report. All you need to do is just click on the download link and get it.
Nanoparticles PPT and Seminar Free Download
Nanoparticles PDF Report Free Download
It was all about Nanoparticles Seminar and PPT with PDF Report. If you liked it then please share it or if you want to ask anything then please hit comment button.


Leave a Reply