PPT: Chromatography | PDF | PowerPoint Presentation Free
PPT: Chromatography PDF | PowerPoint Presentation: Few materials look homogenous but in real they are the combination of distinct substances. A very good example of such thing is the green plants as they have a mixture of distinct pigments. Now, coming to the chromatography then it is a technique which is availed by the scientist for separating the inorganic compounds and organic compounds. The separation of organic compounds and inorganic compounds are done for the purpose of analyzing and studying the compounds. The process of analyzing the compound aids the scientist to picture out the substances that form the compound.
Also See: HPLC PPT
PPT: Chromatography | PDF | PowerPoint Presentation
So, we can say that chromatography is a physical technique for observing solvents and mixtures. Color writing is the meaning of the term chromatography and it is a method in which a chemist can test the mixtures of liquid. Chromatography was invented in 1903 by a Russian botanist and this happened when he was studying plant life’s coloring materials. The name of the Russian scientist was M.S.Tswett.
Chromatography is a very vital method and two chromatographers have been honored with the Nobel prizes. At present, in the world, more than 60% of chemical analysis is done by availing chromatography and by the variation of chromatography. It is availed in many distinct ways and few people avail it to know the inside condition of a solid or a liquid. At the same time, this method is availed in determining the unknown substances. The detectives, F.B.I and Police people avail it at the time of solving a crime. Not only in this but also it is availed in to decide or settle the following things:
- Lead in water
- PCB’s in fish
- Cocaine in urine
- Alcohol in blood
Also See: IR spectroscopy ppt
Chromatography is availed by distinct people in distinct ways and differential migration is the thing on which it is based. In a mobile phase, solutes go via a stationary phase. Solutes with more affinity for the mobile phase stay for more time in the mobile phase when compared with the solutes that go to stationary phase. The solutes get separate as they move via stationary phase and this process is known as chromatography development.
Working of Chromatography:
There are two phases in a chromatography and they are as below:
- Mobile phase
- Stationary phase
The mobile phase is a phase that moves and the stationary phase is a phase which remains stationary. The compounds which have to be tested are picked up by the mobile phase when it goes through the stationary phase which is nothing but the stationary phase. As we know that the behavior of mobile phase is to move and it collects the compounds when it moves through the stationary phase. In a stationary phase, at distinct points the distinct components of a compound are absorbed and also the distinct components prevent moving with mobile phase. This is the procedure by which the results of chromatography are achieved.
Also See: Non Technical Topics
The mobile phase acts as a solvent in the paper and thin-layer chromatography. A piece of paper is placed in a solvent and that acts as a stationary phase in paper chromatography. A thin-layer cell plays the role of stationary phase in thin-layer chromatography. These kinds of chromatography avail capillary action to travel the solvent via the stationary phase.
Retention factor (Rf):
- The retention factor is denoted by Rf and is a quantitative indication that tells about the distance traveled by a particular compound in a particular solvent.
- This value helps to know that the known compound and unknown compound are similar or not. If the value of Rf of a known compound is equal to the Rf value of an unknown compound then it means that the two compounds are the same or identical.
- Retention factor is defined as a ratio of distance traveled by the solute to the distance traveled by the solvent front.
- Rf = D1/D2
- Rf = distance traveled by solute/ distance traveled by the solvent front
The diagrammatic representation of retention factor is as explained below:
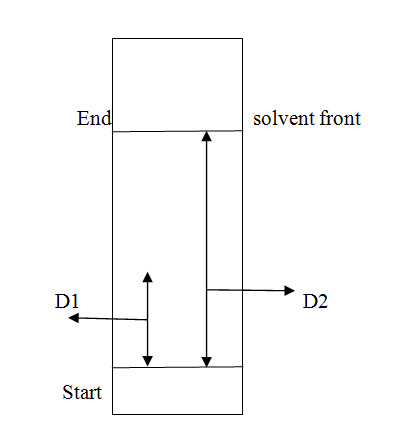
Fig1: Explaining Retention Factor
Also See: Lipids PPT
Types of Chromatography:
The distinct types of chromatography are as explained below:
- Liquid chromatography
- Gas chromatography
- Thin-layer chromatography
- Paper chromatography
The below table explains the chromatography and its applications in the real world and it is as follows:
| Chromatography | Application |
| Liquid chromatography | It is used in testing the water samples to know the pollution. |
| Gas chromatography | This type of chromatography is availed in the forensics lab to compare the fibers which are found on a victim body, detect bombs in airports and also used in identifying and quantifying the drugs like alcohol. |
| Thin-layer chromatography | It is availed in identifying the pesticides in the food and also used to work on the dye composition of fibers in the forensics laboratory. |
| Paper chromatography | It is availed in RNA fingerprinting and also in separating anions and amino acids. |
Fig2: Table about Chromatography and its Applications
Also See: Hypothesis ppt
Content of the Chromatography PPT and PDF
- Introduction to chromatography
- History
- Principles
- Importance
- Chromatographic terms
- Classification of chromatography
- Adsorption chromatography
- Partition chromatography
- Gas-liquid phase chromatrography
- Solid-liquid phase chromatrography
- Liquid-gas phase chromatrography
- Liquid-liquid phase chromatrography
- Important properties of liquid phase
- Conclusion
Here we are giving you Chromatography PPT with PDF. All you need to do is just click on the download link and get it.
Chromatography PDF Free Download
It was all about Chromatography PPT with PDF. If you liked it then please share it or if you want to ask anything then please hit the comment button.



I want ppt for distillation and liquid liquid extraction