IR spectroscopy PPT | PDF For Free Download
IR spectroscopy PPT | PDF For Free Download: IR spectroscopy or infrared spectroscopy is also called as vibrational spectroscopy. It implicates an interaction of infrared radiation with matter. Absorption spectroscopy techniques are covered in it and are used to recognize chemicals and study chemicals. An infrared spectrometer is a tool with which the infrared spectroscopy can be done, an infrared spectrometer is also called as a spectrophotometer and is availed to generate an infrared spectrum.
Also See: LIPIDS PPT and PDF
IR spectroscopy PPT | PDF For Free Download
The infrared spectrum is fundamentally a graph which has thefrequency on a horizontal axis or else wavelength on the horizontal axis and infrared light absorbance on vertical axis. Reciprocal centimeters or wave numbers are the units of frequency availed in IR spectra. The microns or micrometers are units used to represent IR wavelength. A usual equipment of laboratory which avails this method is a Fourier transform infrared spectrometer and also called as FTIR spectrometer. The diagrammatic representation of IR spectrometer is as below:
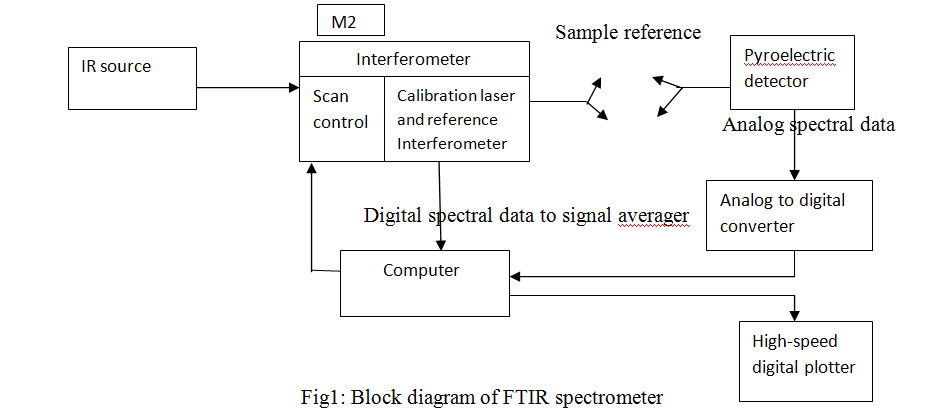
In the electromagnetic spectrum, the portion of infrared is divided into the below regions and they are as follows:
- Near-infrared region
- Mid-infrared region
- Far-infrared region
Also See: HPLC PPT
They are named so because of their relationship with the visible spectrum. The classifications of all these regions are based on the properties of electromagnetic or relative molecular.
- Near-infrared region: The near-infrared region has a frequency of 3.8 * 10^14 – 1.2 * 10^14 Hz, wave numbers of 12800 – 4000 cm^-1, and wavelength of 0.8 – 2.5 µm. It can excite harmonic vibrations and overtone vibrations.
- Mid-infrared region: The mid-infrared region has a frequency of 3.8 * 10^14 – 1.2 * 10^14 Hz, wave numbers of 4000 – 200 cm^-1 and wavelength of 2.5 – 50 µm and is availed for studying the structure of associated rotational vibration and also the fundamental vibrations.
- Far-infrared region: The region of far-infrared has a frequency of 3.8 * 10^14 – 1.2 * 10^14 Hz, wave numbers of 200 – 10 cm^-1, and wavelength of 50-100µm and are availed for rotational spectroscopy.
- Most used region: This region has a frequency of 3.8 * 10^14 – 1.2 * 10^14 Hz, wave numbers of 4000 – 670 cm^-1 and wavelength of 2.5 – 15 µm.
Among many spectroscopic techniques, IR spectroscopy is a widely availed method because the groups which absorb in IR region engross within a particular region of wavelength. The peaks of absorption in an ultraviolet region and visible region are less sharp when compared with the absorption peaks of this region.
Distinct functional groups absorb distinct IR radiation frequency and because of this, determining the functional groups becomes very sensitive in infrared spectroscopy. In organic compounds and in inorganic compounds, IR spectroscopy plays a vital role in identifying distinct substances and also in the structure analysis of the substances. It is also availed in the mixtures of similar compounds which are complex for the qualitative analysis and also for the quantitative analysis.
Also See: Chromatography ppt
Theory of IR spectroscopy:
Infrared spectroscopy takes an unfair advantage of the truth that molecules suck up frequencies and this nature of absorbing is a characteristic of the molecule’s structure. This taking up of the frequencies happens at a resonant frequency, a resonant frequency is a frequency where vibrational frequency matches absorbed radiation.
Infrared energy modes:
Infrared active modes are the stretching vibrational modes, bending vibrational modes and these motions produce the absorptions. The occurrence of asymmetric stretching vibrations is high at the higher frequencies than the occurrence of symmetric stretching vibrations. Similarly, at high frequencies stretching vibrations occur than the bending vibrations.
Influence factors of IR:
The influence factors of IR are as follows:
- Isotope effects
- Solvent effects
Also See: Researched Methodology PPT and PDF
Advantages of IR:
The advantages of IR as below:
- High scan speed
- High resolution
- High sensitivity
- Wide range of applications
- Large amount of information
- Non-destructive
Disadvantages of IR:
The disadvantages of IR are as below:
- Sample constraint
- Spectrum complication
- Quantification
Also See: Non Technical Seminar Topics
Content of the IR spectroscopy PPT and PDF
- What is Infrared Spectroscopy?
- IR Spectroscopic Process
- Uses and Application
- Advantages
- Disadvantages
- Conclusion
- Reference
Here we are giving you IR spectroscopy PPT with PDF. All you need to do is just click on the download link and get it.
It was all about IR spectroscopy PPT with PDF. If you liked it then please share it or if you want to ask anything then please hit comment button.



Leave a Reply